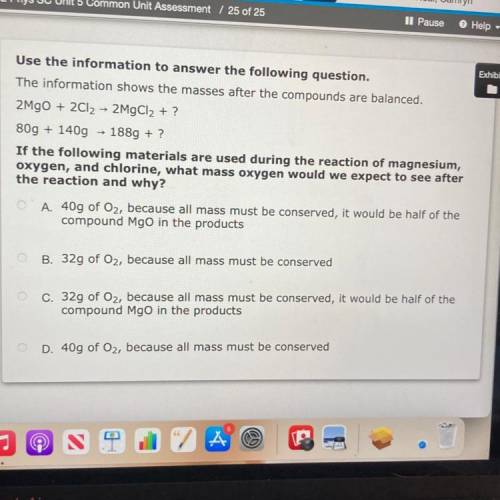
The information shows the masses after the compounds are balanced.
2MgO + 2Cl2 → 2MgCl2 + ?
8...

Chemistry, 11.03.2021 22:30 luthfipadasseri
The information shows the masses after the compounds are balanced.
2MgO + 2Cl2 → 2MgCl2 + ?
80g + 140g + 1889 + ?
If the following materials are used during the reaction of magnesium,
oxygen, and chlorine, what mass oxygen would we expect to see after
the reaction and why?
O A. 40g of Oz, because all mass must be conserved, it would be half of the
compound Mgo in the products
O B. 32g of Oz, because all mass must be conserved
c. 32g of Oz, because all mass must be conserved, it would be half of the
compound Mgo in the products
D. 40g of Oz, because all mass must be conserved

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:10
Describe the difference between a. a hypothesis and a theory and b. an observation and an experiment.
Answers: 1


Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

You know the right answer?
Questions

Mathematics, 12.08.2021 04:30


Social Studies, 12.08.2021 04:30





Physics, 12.08.2021 04:30

Business, 12.08.2021 04:30




Mathematics, 12.08.2021 04:30

Physics, 12.08.2021 04:30


English, 12.08.2021 04:30


Mathematics, 12.08.2021 04:30


History, 12.08.2021 04:40



