35 POINTS
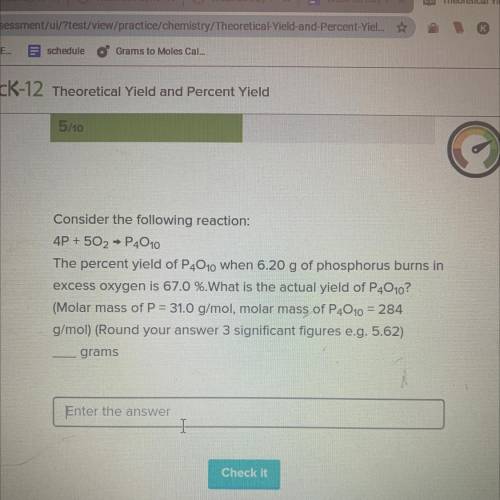
Consider the following reaction:
4P + 502 → P4010
The percent yield of P4010...

Chemistry, 11.03.2021 19:30 dontcareanyonemo
35 POINTS
Consider the following reaction:
4P + 502 → P4010
The percent yield of P4010 when 6.20 g of phosphorus burns in
excess oxygen is 67.0 %.What is the actual yield of P4010?
(Molar mass of P = 31.0 g/mol, molar mass of P4010 = 284
g/mol) (Round your answer 3 significant figures e. g. 5.62)
grams

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

You know the right answer?
Questions




Mathematics, 19.07.2019 02:00



Mathematics, 19.07.2019 02:00


Mathematics, 19.07.2019 02:00

History, 19.07.2019 02:00

Mathematics, 19.07.2019 02:00


Mathematics, 19.07.2019 02:00



Biology, 19.07.2019 02:00


Biology, 19.07.2019 02:00

Mathematics, 19.07.2019 02:00



