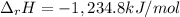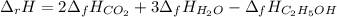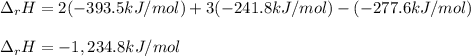Alcohol is used as a source of fuel for some automobiles. The standard molar enthalpy of formation of ethanol C2H5OH(l) is -277.6 kJ/mol. For the balanced reaction equation of ethanol use:
C2H5OH(l) + 3O2 --> 2CO2 + 3H2O.
Ethanol is a liquid, but all the other chemicals in this reaction are gases. What is the enthalpy change of this reaction in kJ/mol?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:50
2points why do scientists need governmental funding? o a. government politicians ask all the important scientific questions. o b. scientists have to pay taxes to the government on the money they make. o c. the cost of doing scientific research can be very high. o d. the government is controlled by scientists. submit
Answers: 3

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3


Chemistry, 23.06.2019 07:30
Can you guys answer these questions i need it before 1: 00pm
Answers: 3
You know the right answer?
Alcohol is used as a source of fuel for some automobiles. The standard molar enthalpy of formation o...
Questions


Physics, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50


Mathematics, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50


Chemistry, 12.12.2020 15:50




History, 12.12.2020 15:50



Mathematics, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50