Chemistry, 10.03.2021 20:00 chenepiernas
Washing soda is a form of a hydrated sodium carbonate (Na2CO3 ∙ 10H2O). If a 10g sample was heated until all the water was driven off and only 3.65 g of anhydrous sodium carbonate (106 g/mol) remained, what is the percent error in obtaining the anhydrous sodium carbonate?
Na2CO3 ∙ 10H2O → Na2CO3 + 10H2O
a
0.16%
b
1.62%
c
3.65%
d
2.51%
please help

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d.the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 21.06.2019 22:30
Which feature do highland climates have that lower elevation areas do not?
Answers: 1

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1
You know the right answer?
Washing soda is a form of a hydrated sodium carbonate (Na2CO3 ∙ 10H2O). If a 10g sample was heated u...
Questions


SAT, 01.12.2021 02:30





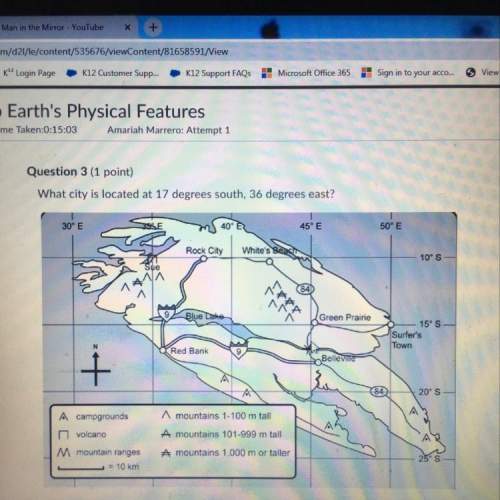
Mathematics, 01.12.2021 02:30



Mathematics, 01.12.2021 02:30







Medicine, 01.12.2021 02:30

Chemistry, 01.12.2021 02:30

Computers and Technology, 01.12.2021 02:30

Computers and Technology, 01.12.2021 02:30