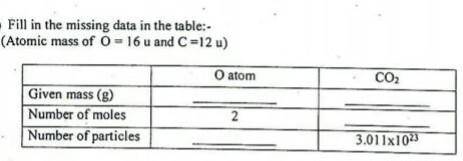
Answer this with steps class 9
...

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

You know the right answer?
Questions

Mathematics, 10.07.2019 14:30


Computers and Technology, 10.07.2019 14:30

Mathematics, 10.07.2019 14:30


Mathematics, 10.07.2019 14:30

Mathematics, 10.07.2019 14:30

Physics, 10.07.2019 14:30


Mathematics, 10.07.2019 14:30


Mathematics, 10.07.2019 14:30


English, 10.07.2019 14:30


Physics, 10.07.2019 14:30

Mathematics, 10.07.2019 14:30

History, 10.07.2019 14:30


Mathematics, 10.07.2019 14:30