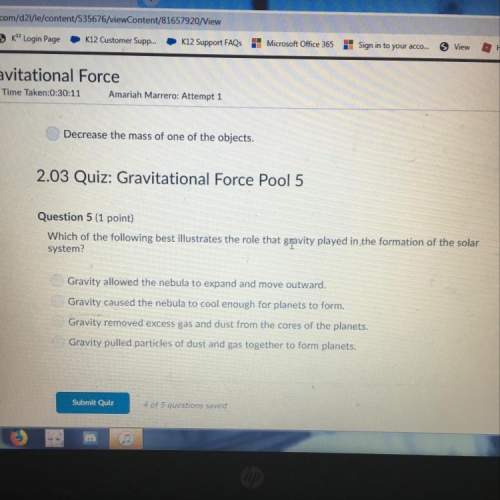
(the * is a multiplication sign btw)
Calculate the molar mass of the compound AI(NO3)3*9H20....

Chemistry, 10.03.2021 16:40 madison1284
(the * is a multiplication sign btw)
Calculate the molar mass of the compound AI(NO3)3*9H20.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:00
The most efficient way to establish the best possible economizer position is to measure
Answers: 1

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2


Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Questions

Physics, 02.10.2020 15:01




History, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Chemistry, 02.10.2020 15:01

English, 02.10.2020 15:01

World Languages, 02.10.2020 15:01

Physics, 02.10.2020 15:01


Physics, 02.10.2020 15:01




History, 02.10.2020 15:01



Mathematics, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01