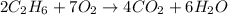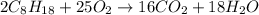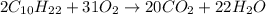Kindly Help Please!
Can you write the symbol equations for each of the reactions below:
and C...

Chemistry, 08.03.2021 14:00 montanolumpuy
Kindly Help Please!
Can you write the symbol equations for each of the reactions below:
and Can you balance them?
1) Ethane + Oxygen = Carbon Dioxide + Water
2) Butane + Oxygen = Carbon Dioxide + Water
3) Octane + Oxygen = Carbon Dioxide + water
4) Decane + Oxygen = Carbon Dioxide + Water
thank you!

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Acylinder is filled with 2.00 moles of nitrogen, 3.00 moles of argon and 5.00 moles of helium. if the gas mixture is at stp, what is the partial pressure of the argon
Answers: 1

Chemistry, 22.06.2019 19:20
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3

Chemistry, 23.06.2019 06:00
What volume of 0.500 mol/l hydrochloric acid, hci (aq) is required to react completely with 1.00 g of aluminum hydroxide, ai(oh)3 (s)?
Answers: 1

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2
You know the right answer?
Questions



Mathematics, 30.03.2020 21:04









Law, 30.03.2020 21:05



Mathematics, 30.03.2020 21:05





English, 30.03.2020 21:05