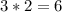Observe this chemical reaction:
4 Fe + 302- 2Fe 203
How many atoms of oxygen are in thi...

Chemistry, 07.03.2021 18:30 imknutson962
Observe this chemical reaction:
4 Fe + 302- 2Fe 203
How many atoms of oxygen are in this !

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 03:00
Which of the dna typing techniques do you think you would choose if you had to analyze a dna sample? why?
Answers: 1

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3
You know the right answer?
Questions

Biology, 04.10.2019 22:00


English, 04.10.2019 22:00




Social Studies, 04.10.2019 22:00

Mathematics, 04.10.2019 22:00

History, 04.10.2019 22:00


Mathematics, 04.10.2019 22:00


History, 04.10.2019 22:00


Mathematics, 04.10.2019 22:00

English, 04.10.2019 22:00

Mathematics, 04.10.2019 22:00

Chemistry, 04.10.2019 22:00