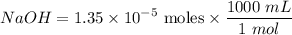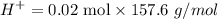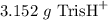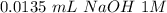Chemistry, 05.03.2021 06:00 naomicervero
Tris is a molecule that can be used to prepare buffers for biochemical experiments. It exists in two forms: Tris (a base) and TrisH (an acid). The MW of Tris base is 121.14 g/mol; the MW of TrisH is 157.6 g/mol (the extra weight is due to the Cl- counterion that is present in the acid). The Ka of the acid is 8.32 X 10-9. Assume that you have TrisH in solid form (a powder), unlimited 1M HCl, unlimited 1 M NaOH and an unlimited supply of distilled water. How would you prepare 1 L of a 0.02 M Tris Buffer, pH

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2
You know the right answer?
Tris is a molecule that can be used to prepare buffers for biochemical experiments. It exists in two...
Questions

History, 15.12.2020 01:00


Social Studies, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00


Biology, 15.12.2020 01:00

Biology, 15.12.2020 01:00


Mathematics, 15.12.2020 01:00


Biology, 15.12.2020 01:00


Spanish, 15.12.2020 01:00

History, 15.12.2020 01:00



History, 15.12.2020 01:00

English, 15.12.2020 01:00


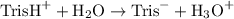
![$K_a = \frac{[\text{Tris}^-]\times[\text{H}_3\text{O}^-]}{[\text{TrisH}^+]}$](/tpl/images/1170/9735/4efa1.png)
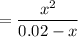
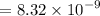
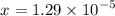 moles of acid
moles of acid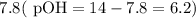 , we must have an
, we must have an 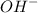 concentration of
concentration of ![$[OH^-] = 10^{-pOH}$](/tpl/images/1170/9735/e3cb0.png)
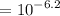
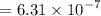 moles of base
moles of base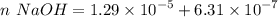
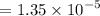 moles
moles