
Chemistry, 05.03.2021 03:40 eprice32315
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. An industrial chemist studying this reaction fills a flask with of sulfur dioxide gas and of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of sulfur trioxide gas to be . Calculate the pressure equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. Round your answer to significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
An empty fuel tank can still contain and therefore can be even more dangerous than one full of liquid fuel.
Answers: 1

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 22:30
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1

Chemistry, 23.06.2019 02:00
To calculate the molarity of a solution, you need to know the moles of solute and the
Answers: 2
You know the right answer?
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid...
Questions

Mathematics, 11.10.2019 19:30


Biology, 11.10.2019 19:30

Mathematics, 11.10.2019 19:30



Business, 11.10.2019 19:30


Biology, 11.10.2019 19:30


History, 11.10.2019 19:30


History, 11.10.2019 19:30



Mathematics, 11.10.2019 19:30

Mathematics, 11.10.2019 19:30


Spanish, 11.10.2019 19:30

 "
"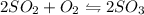

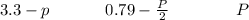
![p= 0.47 \ \ atm\\\\SO_2=3.3-0.47 = 2.83 \ \ atm\\\\O_2= 0.74 -\frac{0.47}{2}=0.74-0.235=0.555 \ atm\\\\K_P=\frac{[PSO_3]^2}{[PSO_2]^2[PO_2]}\\\\](/tpl/images/1170/6349/28d0a.png)




