
A buffer solution contains 0.345 M acetic acid and 0.377 M sodium acetate . If 0.0613 moles of potassium hydroxide are added to 250 mL of this buffer, what is the pH of the resulting solution ? (Assume that the volume does not change upon adding potassium hydroxide. )

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2

Chemistry, 23.06.2019 07:50
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1
You know the right answer?
A buffer solution contains 0.345 M acetic acid and 0.377 M sodium acetate . If 0.0613 moles of potas...
Questions

Mathematics, 04.06.2021 01:00



Mathematics, 04.06.2021 01:00


Mathematics, 04.06.2021 01:00






History, 04.06.2021 01:00

English, 04.06.2021 01:00

Mathematics, 04.06.2021 01:00

Mathematics, 04.06.2021 01:00


Chemistry, 04.06.2021 01:00



Mathematics, 04.06.2021 01:00

![\frac{[CH_3COO^-]}{[CH_3COOH]}](/tpl/images/1163/7311/4fc64.png)
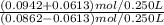 = 5.54
= 5.54

