
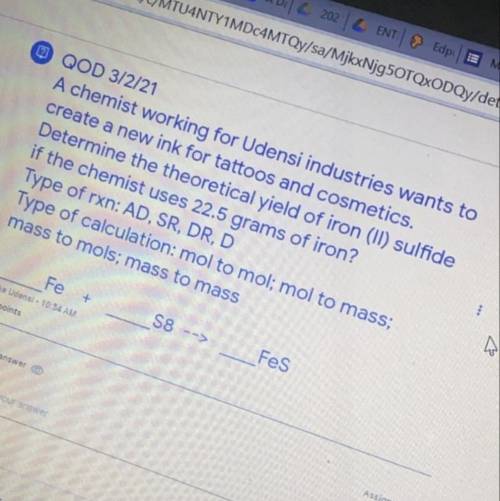
A chemist working for Udensi industries wants to create a new ink for tattoos and cosmetics. Determine the theoretical yield of iron (ll) sulfide if the chemist uses 22.5 grams of iron ? Type of rxn : AD , SR, DR , D Type of calculation : mol to mol ; mol to mass ; mass to mols mass to mass Fe S8--> Fes

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3

Chemistry, 23.06.2019 10:00
Two moles of potassium chloride and three moles of oxygen are produced from the decomposition of two moles of potassium chlorate, kcos3(s). write the balanced equation. how many moles of oxygen are produced from 12 moles of potassium chlorate
Answers: 1

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
You know the right answer?
A chemist working for Udensi industries wants to create a new ink for tattoos and cosmetics. Determi...
Questions


Mathematics, 04.12.2019 02:31


Biology, 04.12.2019 02:31

Mathematics, 04.12.2019 02:31

Computers and Technology, 04.12.2019 02:31

History, 04.12.2019 02:31


Geography, 04.12.2019 02:31




Social Studies, 04.12.2019 02:31


Health, 04.12.2019 02:31


History, 04.12.2019 02:31





