
Chemistry, 01.03.2021 22:10 naomicervero
Suppose you want to prepare a buffer with a pH of 4.59 using formic acid. What ratio of [sodium formate]/[formic acid) do you need to make this buffer? Formic acid has a K, of 1.8x10 4 Enter your answer to three significant figures.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
Suppose you want to prepare a buffer with a pH of 4.59 using formic acid. What ratio of [sodium form...
Questions


Mathematics, 08.10.2020 01:01




Arts, 08.10.2020 01:01

Computers and Technology, 08.10.2020 01:01




Health, 08.10.2020 01:01





Advanced Placement (AP), 08.10.2020 01:01




Spanish, 08.10.2020 01:01

![\frac{[A^-]}{[HA]}](/tpl/images/1158/1330/1d212.png)
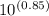 =
=

