Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3


Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
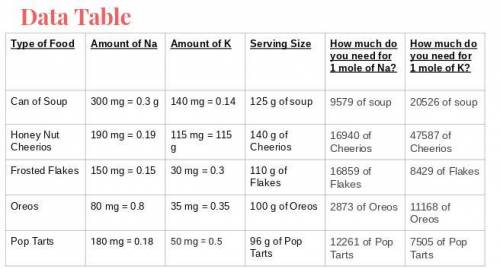
What food item will provide a minimum of 1 mole of both sodium and potassium with the least mass? Wh...
Questions

History, 27.05.2020 09:58

Mathematics, 27.05.2020 09:58


Mathematics, 27.05.2020 09:58


Mathematics, 27.05.2020 09:58

Mathematics, 27.05.2020 09:58

Mathematics, 27.05.2020 09:58

History, 27.05.2020 09:58

Mathematics, 27.05.2020 09:58


Mathematics, 27.05.2020 09:58


Chemistry, 27.05.2020 09:58






Mathematics, 27.05.2020 09:58