
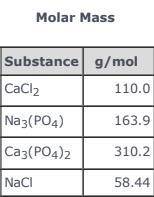
3CaCl2(aq) + 2Na3(PO4)(aq) → Ca3(PO4)2(s)+ 6NaCl(aq) Use the balanced equation and the Molar Mass table above to answer the following question. How much Ca3(PO4)2(s) could be produced in an industrial process if 55.00 g of CaCl2 in solution reacted completely with sufficient Na3(PO4)(aq)?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:20
Which type of substance ionizes partially and gives off hydrogen ions when dissolved in water? a. strong acid b. strong base c. weak acid d. weak base
Answers: 1

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1

You know the right answer?
3CaCl2(aq) + 2Na3(PO4)(aq) → Ca3(PO4)2(s)+ 6NaCl(aq) Use the balanced equation and the Molar Mass ta...
Questions

Physics, 03.10.2021 14:00


Biology, 03.10.2021 14:00


Advanced Placement (AP), 03.10.2021 14:00

Biology, 03.10.2021 14:00

Chemistry, 03.10.2021 14:00


Law, 03.10.2021 14:00

Social Studies, 03.10.2021 14:00

Mathematics, 03.10.2021 14:00

History, 03.10.2021 14:00



Physics, 03.10.2021 14:00


Spanish, 03.10.2021 14:00

History, 03.10.2021 14:00

English, 03.10.2021 14:00



