How do I find the mass for the pumpkin what scale do I put it in and what do I do?
...

Chemistry, 24.02.2021 16:00 nananunu2747
How do I find the mass for the pumpkin what scale do I put it in and what do I do?
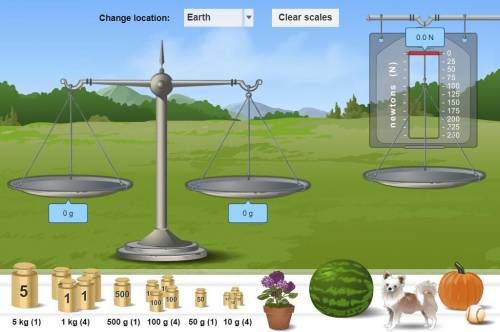

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1
You know the right answer?
Questions


Business, 18.01.2021 14:00


Mathematics, 18.01.2021 14:00

Biology, 18.01.2021 14:00

Physics, 18.01.2021 14:00

English, 18.01.2021 14:00




English, 18.01.2021 14:10


Mathematics, 18.01.2021 14:10

Social Studies, 18.01.2021 14:10

Social Studies, 18.01.2021 14:10

Chemistry, 18.01.2021 14:10

Computers and Technology, 18.01.2021 14:10

Mathematics, 18.01.2021 14:10

Mathematics, 18.01.2021 14:10



