
Chemistry, 23.02.2021 16:40 monaae3824
I'LL MARK YOU BRAINLIEST! Please help me balance these chemical reactions. (I'M NOT IN COLLEGE, I'M IN 10TH GRADE) Please help me
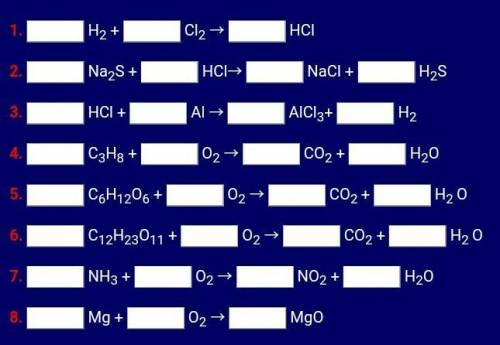

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 17:00
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2

Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2

Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2

Chemistry, 22.06.2019 22:30
Why is it possible for different microorganisms to extract energy not only from carbohydrates and other biological molecules but from a large variety of substances?
Answers: 1
You know the right answer?
I'LL MARK YOU BRAINLIEST! Please help me balance these chemical reactions. (I'M NOT IN COLLEGE, I'M...
Questions

Mathematics, 01.07.2019 04:30



Social Studies, 01.07.2019 04:30


Social Studies, 01.07.2019 04:30

History, 01.07.2019 04:30

Mathematics, 01.07.2019 04:30

Social Studies, 01.07.2019 04:30


History, 01.07.2019 04:30




Advanced Placement (AP), 01.07.2019 04:30

Chemistry, 01.07.2019 04:30




History, 01.07.2019 04:30



