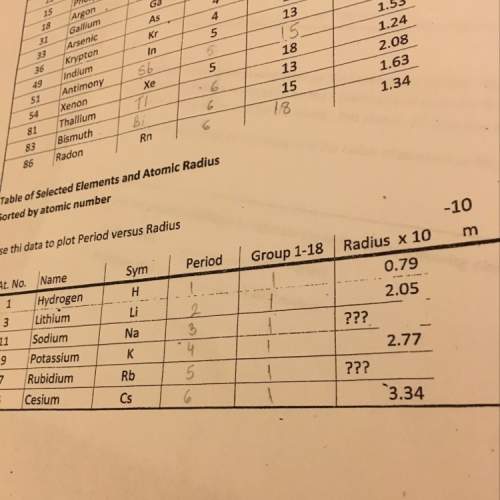
compute the theoretical yield of the product (in

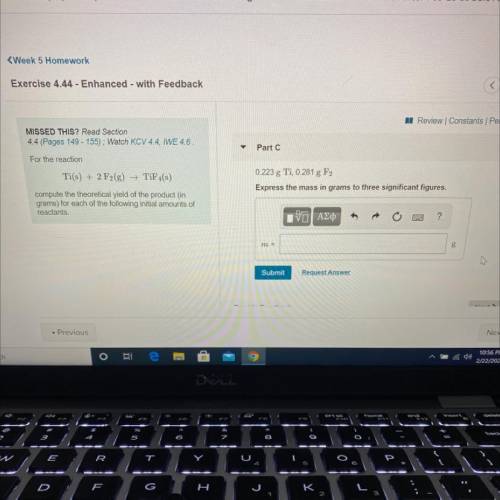
For the reaction
Ti(s) + 2 F2(g) →TiF4(s)
compute the theoretical yield of the product (in
grams) for each of the following initial amounts of
reactants.
0.223g Ti, 0.281g F2
Express your answer using three significant figures.
please help! will give brainliest.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2

You know the right answer?
For the reaction
Ti(s) + 2 F2(g) →TiF4(s)
compute the theoretical yield of the product (in
compute the theoretical yield of the product (in
Questions





Biology, 04.12.2021 01:10


Computers and Technology, 04.12.2021 01:10


Mathematics, 04.12.2021 01:10

Mathematics, 04.12.2021 01:10

Chemistry, 04.12.2021 01:10


Mathematics, 04.12.2021 01:10




Biology, 04.12.2021 01:10

Mathematics, 04.12.2021 01:10

Physics, 04.12.2021 01:10