
Chemistry, 18.02.2021 21:00 larissa1013
suppose you start out with only reactants in a rigid container. if the initial concentration of SO2Cl2(g) is 0.543 M, and 43.6% of this initial concentration remains when the system has reached equilibrium, what are the equilibrium concentrations of each gas in the system

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1

Chemistry, 23.06.2019 06:00
What physical property of gold makes panning a useful way to get gold from streams?
Answers: 2

Chemistry, 23.06.2019 08:00
At 35.0°c and 3.00 atm pressure, a gas has a volume of 1.40 l. what pressure does the gas have at 0.00°c and a volume of 0.950 l? which equation should you use? p2= p1v1t2/t1v2what is the pressure of the gas? 3.92 atm these are the answers
Answers: 1
You know the right answer?
suppose you start out with only reactants in a rigid container. if the initial concentration of SO2C...
Questions





Mathematics, 18.09.2019 20:30


History, 18.09.2019 20:30



History, 18.09.2019 20:30


English, 18.09.2019 20:30

Computers and Technology, 18.09.2019 20:30



Chemistry, 18.09.2019 20:30

Mathematics, 18.09.2019 20:30



Health, 18.09.2019 20:30

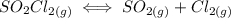
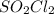 that is being reacted is:
that is being reacted is: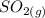 +
+ 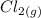
![K_c = \dfrac{[SO_2] [Cl_{2}]}{[SO_2Cl_2]}](/tpl/images/1128/3654/65f33.png)
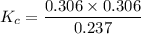
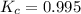
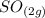 = 0.306 M
= 0.306 M

