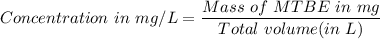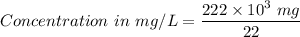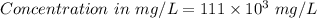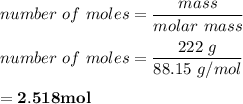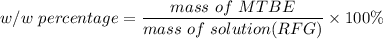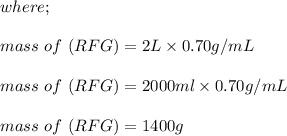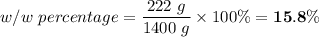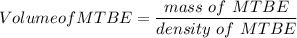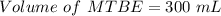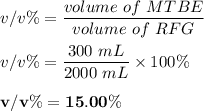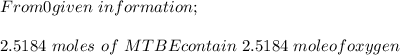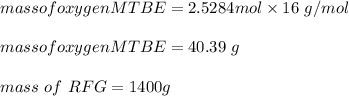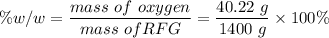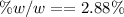Chemistry, 18.02.2021 20:40 QueenBlumple4443
222 g of MTBE (CO(CH3)4) are added to gasoline, resulting in a total volume of 2 L of reformulated gas (RFG). Assume that the density of RFG is 0.70 g/mL, and the density of MTBE is 0.74 g/mL. For a-d, determine the concentration of MTBE in the RFG in the following units?
a. mg/L
b. moles/L
c. % (w/w)
d. % (v/v)
e. What is the % (w/w) oxygen in the RFG due to MTBE?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3
You know the right answer?
222 g of MTBE (CO(CH3)4) are added to gasoline, resulting in a total volume of 2 L of reformulated g...
Questions


Mathematics, 11.01.2021 19:20

Mathematics, 11.01.2021 19:20

Mathematics, 11.01.2021 19:20

Mathematics, 11.01.2021 19:20


Computers and Technology, 11.01.2021 19:20


Mathematics, 11.01.2021 19:20


English, 11.01.2021 19:20


Mathematics, 11.01.2021 19:20

Social Studies, 11.01.2021 19:20

Mathematics, 11.01.2021 19:20