Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3

Chemistry, 22.06.2019 08:40
What is the value of keq for the reaction expressed in scientific notation?
Answers: 1

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
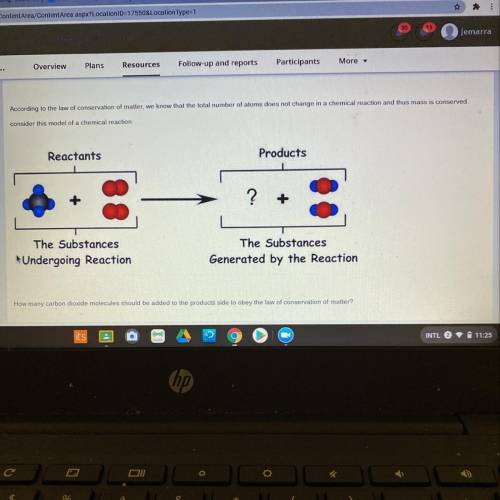
How many carbon dioxide molecules should be added to the products side to obey the law of conservati...
Questions


History, 01.08.2019 23:00

Advanced Placement (AP), 01.08.2019 23:00




Computers and Technology, 01.08.2019 23:00

Mathematics, 01.08.2019 23:00




English, 01.08.2019 23:00


Social Studies, 01.08.2019 23:00



Biology, 01.08.2019 23:00

Computers and Technology, 01.08.2019 23:00


Computers and Technology, 01.08.2019 23:00