
Chemistry, 18.02.2021 01:00 burritomadness
How do I make the molar masses for 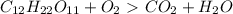 (reactant and products) equal? Please, tell me the steps and how to recalculate.
(reactant and products) equal? Please, tell me the steps and how to recalculate.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:10
How many grams of naoh are needed to make 0.250 liter of a 0.500 m solution of naoh? 0.125 g 5.00 g 2.00 g
Answers: 1

Chemistry, 22.06.2019 23:40
What energy conversion occurs when a sling shot is used to shoot a rock across the room? (2 points) question 2 options: 1) stored mechanical energy is converted to mechanical energy. 2) stored mechanical energy is converted to radiant energy. 3) gravitational energy is converted to radiant energy. 4) gravitational energy is converted to mechanical energy.
Answers: 1

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1
You know the right answer?
How do I make the molar masses for (reactant and products) equal? Please, tell me the steps and how...
Questions

English, 29.01.2020 20:55

Geography, 29.01.2020 20:55







History, 29.01.2020 20:55


Mathematics, 29.01.2020 20:55

Advanced Placement (AP), 29.01.2020 20:55

History, 29.01.2020 20:55


Computers and Technology, 29.01.2020 20:55

Social Studies, 29.01.2020 20:55


Mathematics, 29.01.2020 20:55




