
Chemistry, 16.02.2021 03:10 maysahdabest
Carbon monoxide is a colorless, odorless gas that binds irreversibly to hemoglobin in our blood, causing suffocation and death. CO is formed during incomplete combustion of carbon. One way to represent this equilibrium is: CO(g)C(s) 1/2 O2(g) We could also write this reaction three other ways, listed below. The equilibrium constants for all of the reactions are related. Write the equilibrium constant for each new reaction in terms of K, the equilibrium constant for the reaction above.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 22.06.2019 19:00
Suppose that a certain fortunate person has a net worth of $71.0 billion ($7.10×1010). if her stock has a good year and gains $3.20 billion (3.20×109) in value, what is her new net worth?
Answers: 3
You know the right answer?
Carbon monoxide is a colorless, odorless gas that binds irreversibly to hemoglobin in our blood, cau...
Questions

English, 04.09.2020 20:01

Biology, 04.09.2020 20:01


History, 04.09.2020 20:01


Social Studies, 04.09.2020 20:01

Mathematics, 04.09.2020 20:01


Mathematics, 04.09.2020 20:01

Physics, 04.09.2020 20:01





Mathematics, 04.09.2020 20:01

Social Studies, 04.09.2020 20:01


Mathematics, 04.09.2020 20:01

Mathematics, 04.09.2020 20:01

Mathematics, 04.09.2020 20:01

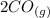 ⇄
⇄ 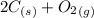
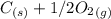 ⇄
⇄ 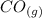 K₂ =
K₂ =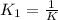
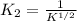
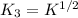
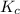 .
.![K=\frac{[O_{2}]}{[CO]^{2}}](/tpl/images/1121/0990/38d27.png)
![K_{1}=\frac{[CO]^{2}}{[O_{2}]}](/tpl/images/1121/0990/e7936.png)
![K_{2}=\frac{[CO]}{[O_{2}]^{1/2}}](/tpl/images/1121/0990/06615.png)
![K_{3}=\frac{[O_{2}]^{1/2}}{[CO]}](/tpl/images/1121/0990/b8d7c.png)
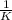 ,
,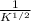 and
and 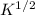 , respectively.
, respectively.

