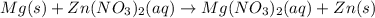Chemistry, 14.02.2021 14:00 kordejah348
What has a complete ionic equation of Mg(s) + Zn2+ + 2NO3 → Mg2+ + 2NO3 + Zn(s)?
O A. Mg(s) + Zn2+ → Mg2+ + Zn(s)
O B. Mg(s) + Zn(NO3)2(aq) + Mg(NO3)2(aq) + Zn(s)
O C. Mg(NO3)2 + Zn(s) → Zn2+ + 2NO3 + Mg(s) O D. Mg(s) + 2Zn(NO3)(aq) + 2MgNO3(aq) + Zn(s)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
What has a complete ionic equation of Mg(s) + Zn2+ + 2NO3 → Mg2+ + 2NO3 + Zn(s)?
O A. Mg(s) + Zn2+...
Questions


Chemistry, 09.12.2020 23:20

Mathematics, 09.12.2020 23:20

Mathematics, 09.12.2020 23:20


Mathematics, 09.12.2020 23:20


Arts, 09.12.2020 23:20


Mathematics, 09.12.2020 23:20


Chemistry, 09.12.2020 23:20


Mathematics, 09.12.2020 23:20





Mathematics, 09.12.2020 23:20

English, 09.12.2020 23:20