
Chemistry, 12.02.2021 08:40 asjessehills2000
The monovalent salt concentration (the predominant solute in the blood cell) for a sample of red blood cells is 0.13 moles/liter. If one of these red blood cells were placed in pure water (at around room temperature, 300 K), and the cell comes to hydrostatic equilibrium with the water, what is the osmotic pressure of the cell (assuming it doesn't burst)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
For the following, determine the type of reaction and then give products.
Answers: 2

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 13:30
Some animals that try to adapt to climate changes eventually die due to starvation, as climate change alters the web.
Answers: 2
You know the right answer?
The monovalent salt concentration (the predominant solute in the blood cell) for a sample of red blo...
Questions

History, 15.07.2019 22:30





Mathematics, 15.07.2019 22:30

Mathematics, 15.07.2019 22:30



History, 15.07.2019 22:30


English, 15.07.2019 22:30


Biology, 15.07.2019 22:30


Mathematics, 15.07.2019 22:30

Mathematics, 15.07.2019 22:30



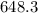 KPa
KPa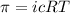
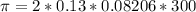
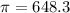 KPa
KPa

