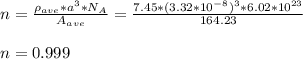Chemistry, 12.02.2021 07:20 senituliii
Some hypothetical alloy is composed of 25 wt% of metal A and 75 wt% of metal B. If the densities of metals A and B are 6.17 and 8.00 g/cm3 , respectively, and their respective atomic weights are 171.3 and 162.0 g/mol, determine whether the crystal structure for this alloy is simple cubic, facecentered cubic, or body-centered cubic. Assume a unit cell edge length of 0.332 nm

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Which best describes why nh4+ can form an ionic bond with ci-?
Answers: 1

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 22.06.2019 23:00
What is formed when amino acids form long chains or polymerize
Answers: 1
You know the right answer?
Some hypothetical alloy is composed of 25 wt% of metal A and 75 wt% of metal B. If the densities of...
Questions


Mathematics, 25.01.2022 01:20

History, 25.01.2022 01:20


English, 25.01.2022 01:20

Mathematics, 25.01.2022 01:20

Mathematics, 25.01.2022 01:20


Mathematics, 25.01.2022 01:20


Chemistry, 25.01.2022 01:20

Mathematics, 25.01.2022 01:20

Mathematics, 25.01.2022 01:20

Computers and Technology, 25.01.2022 01:20

English, 25.01.2022 01:20



English, 25.01.2022 01:20