
Chemistry, 11.02.2021 14:00 abraham1366
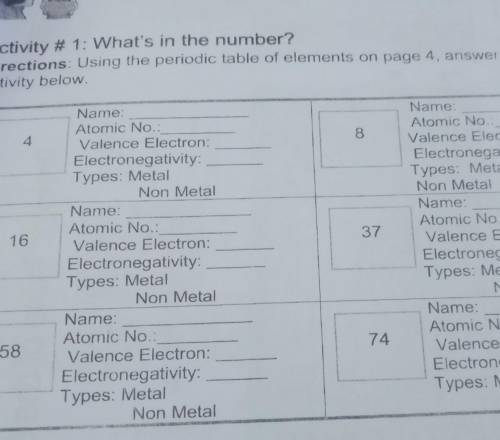
Activity # 1: What's in the number?
Directions: Using the periodic table of elements on page 4, answer the given
activity below
4
8
16
37
Name:
Atomic No.:
Valence Electron:
Electronegativity:
Types: Metal
Non Metal
Name:
Atomic No.:
Valence Electron:
Electronegativity:
Types: Metal
Non Metal
Name:
Atomic No.:
Valence Electron:
Electronegativity:
Types: Metal
Non Metal
Name:
Atomic No.
Valence Electron:
Electronegativity:
Types: Metal
Non Metal
Name:
Atomic No.:
Valence Electron:
Electronegativity:
Types: Metal
Non Metal
Name:
Atomic No.:
Valence Electron:
Electronegativity:
Types: Metal
Non Metal
58
74 pa hellp plsss

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1

Chemistry, 23.06.2019 08:00
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1

Chemistry, 23.06.2019 11:00
Which of the following reactions represents an exothermic reaction? nh3(g) + 12.0 kcal ½n2(g) + 3/2 h2(g) ch4 + 2o2 co2 + 2h2o + 212,800 cal c + 2s cs2, h = 27,550 cal c(graphite) c(diamond), h = 0.45 kcal 2h2o 2h2 + o2, h = +58 kcal
Answers: 1
You know the right answer?
Activity # 1: What's in the number?
Directions: Using the periodic table of elements on page 4, ans...
Questions

Mathematics, 25.05.2021 03:00


Computers and Technology, 25.05.2021 03:00



History, 25.05.2021 03:00

Social Studies, 25.05.2021 03:00

Mathematics, 25.05.2021 03:00

Computers and Technology, 25.05.2021 03:00


Business, 25.05.2021 03:00



Mathematics, 25.05.2021 03:00

Chemistry, 25.05.2021 03:00



Mathematics, 25.05.2021 03:00

Mathematics, 25.05.2021 03:00



