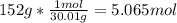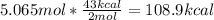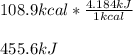Chemistry, 11.02.2021 14:00 santiagoagilg
One mole (mol) of nitrogen monoxide (NO) has a mass of 30.01 g. When
precisely 2 moles of NO(g) are produced in the following chemical reaction, 43
kcal of heat energy is "absorbed."
N2(g) + O2(g) → 2 NO(g), AH = +43 kcal
How much heat (in kJ) is exchanged when 152 g of NO(g) is produced?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
You are to give ampicillin with a recommended dose of 25mg/kg to a child with a mass of 29kg. if stock on hand is 250mg/capsule how many capsules should be given?
Answers: 1

Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 08:30
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 17:20
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2
You know the right answer?
One mole (mol) of nitrogen monoxide (NO) has a mass of 30.01 g. When
precisely 2 moles of NO(g) are...
Questions



Mathematics, 08.11.2019 01:31








Social Studies, 08.11.2019 01:31

Physics, 08.11.2019 01:31


History, 08.11.2019 01:31


World Languages, 08.11.2019 01:31