
Chemistry, 10.02.2021 21:50 kevinh2683
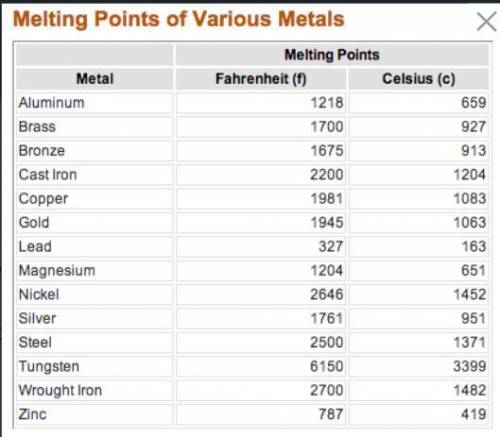
Which metallic substance would require the least amount of thermal energy to melt?
You may need to zoom in on the data table in the pic
A. Gold
B. Nickel
C. Aluminum
D. Steel

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:10
Which statement is true about the part of the electromagnetic spectrum that human eyes can detect? it contains only the colors of the rainbow and television waves. o it is divided into seven ranges of wavelengths. it contains ultraviolet, visible, and infrared light. it is divided into nine ranges of wavelengths.
Answers: 2

Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
Which metallic substance would require the least amount of thermal energy to melt?
You may need to...
Questions




Mathematics, 25.11.2020 07:50

Mathematics, 25.11.2020 07:50

English, 25.11.2020 07:50

Physics, 25.11.2020 07:50


Arts, 25.11.2020 07:50

Mathematics, 25.11.2020 07:50




Mathematics, 25.11.2020 07:50


Health, 25.11.2020 07:50

Mathematics, 25.11.2020 07:50



English, 25.11.2020 07:50



