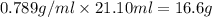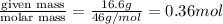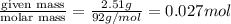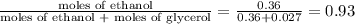Glycerol (C3H8O3), also called glycerine, is widely used in the food and pharmaceutical industries. Glycerol is polar and dissolves readily in water and polar organic solvents like ethanol. Calculate the mole fraction of the solvent in a solution that contains 2.51 g glycerol dissolved in 21.10 mL ethanol (CH3CH2OH; density

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Note the ph and poh values labeled with letters on the ph scale below. based on log rules and the way ph is calculated, what is the difference in [oh– ] concentration between point a and point b. a) 10^1 b) 10^5 c) 10^6 d) 10^7
Answers: 1


Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2
You know the right answer?
Glycerol (C3H8O3), also called glycerine, is widely used in the food and pharmaceutical industries....
Questions

History, 29.06.2021 03:30



Mathematics, 29.06.2021 03:30

Biology, 29.06.2021 03:30

Mathematics, 29.06.2021 03:30

Advanced Placement (AP), 29.06.2021 03:30


History, 29.06.2021 03:30

Mathematics, 29.06.2021 03:30


Social Studies, 29.06.2021 03:30



Mathematics, 29.06.2021 03:30


History, 29.06.2021 03:30

Mathematics, 29.06.2021 03:30

History, 29.06.2021 03:30