The reform reaction between steam and gaseous methane (CH4) produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen. Suppose a chemical engineer studying a new catalyst for the reform reaction finds that 924. liters per second of methane are consumed when the reaction is run at 261.°C and 0.96atm. Calculate the rate at which dihydrogen is being produced.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1

Chemistry, 22.06.2019 04:00
4. absorption has the highest risk of overdose due to increased potency. a. rectal b. oral c. transdermal d. intranasal
Answers: 2

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1
You know the right answer?
The reform reaction between steam and gaseous methane (CH4) produces "synthesis gas," a mixture of c...
Questions

Geography, 29.06.2019 18:30



English, 29.06.2019 18:30




Health, 29.06.2019 18:30

History, 29.06.2019 18:30





Mathematics, 29.06.2019 18:30



Computers and Technology, 29.06.2019 18:30


Mathematics, 29.06.2019 18:30

 ".
".

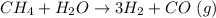
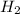 produced:
produced: