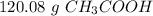Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 23.06.2019 04:31
2ki + pb(no3)2 → 2kno3 + pbi2 determine how many moles of kno3 are created if 0.03 moles of ki are completely consumed.
Answers: 1

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
1.204 x 10^24 molecules of CH3COOH is how many grams?...
Questions



Mathematics, 23.03.2021 20:30

History, 23.03.2021 20:30



Arts, 23.03.2021 20:30



Chemistry, 23.03.2021 20:30

History, 23.03.2021 20:30

Mathematics, 23.03.2021 20:30



Mathematics, 23.03.2021 20:30

English, 23.03.2021 20:30




English, 23.03.2021 20:30

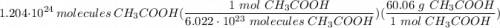 Multiply/Divide:
Multiply/Divide: