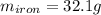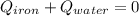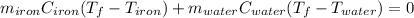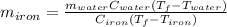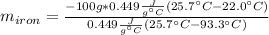Chemistry, 05.02.2021 21:20 eeeeee7891
A sample of iron, which has a specific heat capacity of 0.449 Jg^-1℃^-1, is put into a calorimeter (see sketch at right) that contains 100.0 g of water. The iron sample starts off at container 93.3 °C and the temperature of the water starts off at 22.0 °C. When the temperature of the water stops changing it's 25.7 °C. The pressure remains constant at 1 atm.
Required:
Calculate the mass of the iron sample. Be sure your answer is rounded to 2 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:50
H2so4(aq) + mg(s)—> mgso4(aq) +h2(g) which substance is the acid in the reaction?
Answers: 3

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
A sample of iron, which has a specific heat capacity of 0.449 Jg^-1℃^-1, is put into a calorimeter (...
Questions

Physics, 04.10.2020 03:01

Health, 04.10.2020 03:01




Mathematics, 04.10.2020 03:01

Health, 04.10.2020 03:01




Mathematics, 04.10.2020 03:01



Mathematics, 04.10.2020 03:01


Mathematics, 04.10.2020 03:01


History, 04.10.2020 03:01

History, 04.10.2020 03:01

Chemistry, 04.10.2020 03:01