
Chemistry, 05.02.2021 18:00 kevincaleb1
Use the following equation to answer the question below. How many moles of iron are made from 8.25 moles of Fe2O3?(see pic for equation)
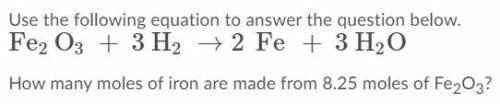

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 23.06.2019 02:00
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1
You know the right answer?
Use the following equation to answer the question below. How many moles of iron are made from 8.25 m...
Questions

Mathematics, 16.12.2020 23:30


Mathematics, 16.12.2020 23:30

Mathematics, 16.12.2020 23:30


Chemistry, 16.12.2020 23:30


Mathematics, 16.12.2020 23:30


English, 16.12.2020 23:30

Mathematics, 16.12.2020 23:30

English, 16.12.2020 23:30

Mathematics, 16.12.2020 23:30





Mathematics, 16.12.2020 23:30


Mathematics, 16.12.2020 23:30



