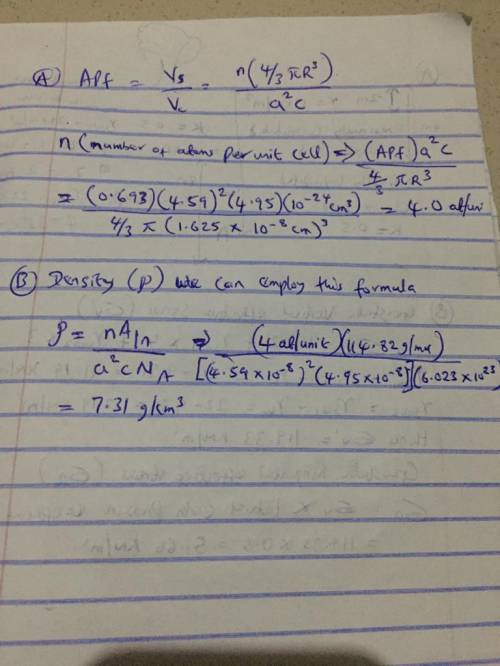Chemistry, 02.02.2021 05:50 ob3ymysins
Indium has a tetragonal unit cell for which the a and c lattice parameters are 0.459 and 0.495 nm, respectively. (a) If the atomic packing factor and atomic radius are 0.693 and 0.1625 nm, respectively, determine the number of atoms in each unit cell. (b) The atomic weight of indium is 114.82 g/mol; compute its theoretical density. (a) Enter your answer for part (a) in accordance to the question statement atoms/unit cell (b) Enter your answer for part (b) in accordance to the question statement g/cm3

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

Chemistry, 23.06.2019 12:00
Which of the following statements is true? a. most heat energy is easily recovered and used for useful actions. b. friction causes molecules to vibrate more slowly. burning air and gasoline in an c. engine changes chemical energy into mechanical energy. it is impossible to d. change mechanical energy into mechanical energy.
Answers: 1
You know the right answer?
Indium has a tetragonal unit cell for which the a and c lattice parameters are 0.459 and 0.495 nm, r...
Questions

English, 05.11.2020 07:40

Chemistry, 05.11.2020 07:40



English, 05.11.2020 07:40


Mathematics, 05.11.2020 07:40




Mathematics, 05.11.2020 07:40


Mathematics, 05.11.2020 07:40

Mathematics, 05.11.2020 07:40

Physics, 05.11.2020 07:40

Mathematics, 05.11.2020 07:40

Mathematics, 05.11.2020 07:40


English, 05.11.2020 07:40