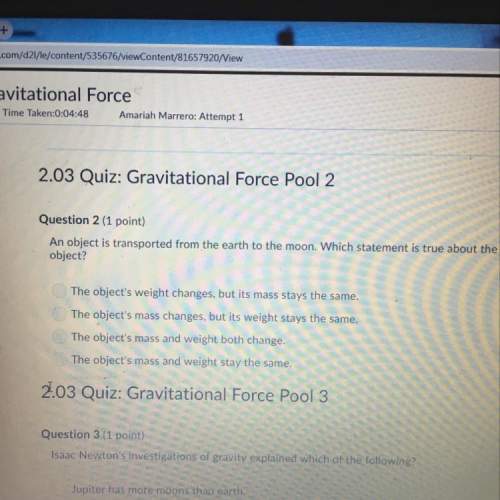
Chemistry, 01.11.2019 21:31 korleone8071
The relative positions of protons, neutrons, and electrons in an atom

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2

Chemistry, 23.06.2019 09:00
What properties would have caused early researchers to name hydrogen "inflammable air”
Answers: 3
You know the right answer?
The relative positions of protons, neutrons, and electrons in an atom...
Questions

Biology, 26.02.2021 04:20


Mathematics, 26.02.2021 04:20


Social Studies, 26.02.2021 04:20


Social Studies, 26.02.2021 04:20



Computers and Technology, 26.02.2021 04:20

Mathematics, 26.02.2021 04:20

Mathematics, 26.02.2021 04:20

English, 26.02.2021 04:20



Social Studies, 26.02.2021 04:20

Business, 26.02.2021 04:20



Mathematics, 26.02.2021 04:20