
Chemistry, 01.02.2021 21:40 cjd1214812148
Problem 3 A sample of 2.37 moles of an ideal diatomic gas experiences a temperature increase of 65.2 K at constant volume. (a) Find the increase in internal energy if only translational and rotational motions are possible. (b) Find the increase in internal energy if translational, rotational, and vibrational motions are possible. (c) How much of the energy calculated in (a) and (b) is translational kinetic energy?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2

Chemistry, 23.06.2019 08:30
Benzonitrile (c6h5cn) is reduced to two different products depending on the reducing agent used. treatment with lithium aluminum hydride followed by water forms k, which has a molecular ion in its mass spectrum at 107 and the following ir absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. treatment with a milder reducing agent forms l, which has a molecular ion in its mass spectrum at 106 and the following ir absorptions: 3086, 2850, 2820, 2736, 1703, and 1600 cm-1. l shows fragments in its mass spectrum at m/z = 105 and 77. propose structures for k and l and choose an explanation for how this could be concluded.
Answers: 3
You know the right answer?
Problem 3 A sample of 2.37 moles of an ideal diatomic gas experiences a temperature increase of 65.2...
Questions

Mathematics, 18.03.2021 22:00

History, 18.03.2021 22:00

Mathematics, 18.03.2021 22:00

Social Studies, 18.03.2021 22:00



History, 18.03.2021 22:00


Mathematics, 18.03.2021 22:00

History, 18.03.2021 22:00


Mathematics, 18.03.2021 22:00

History, 18.03.2021 22:00



English, 18.03.2021 22:00

Mathematics, 18.03.2021 22:00

History, 18.03.2021 22:00

History, 18.03.2021 22:00

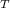 = 1927.06 J
= 1927.06 J

