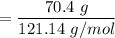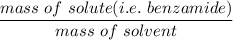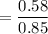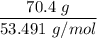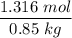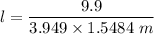Chemistry, 01.02.2021 20:40 alyonaprotopopova
When 70.4 g of benzamide (C7H7NO) are dissolved in 850. g of a certain mystery liquid X, the freezing point of the solution is 2.7 C lower than the freezing point of pure X. On the other hand, when 70.4 g of ammonium chloride (NH CI) are dissolved in the same mass of X, the freezing point of the solution is 9.9 °C lower than the freezing point of pure X.
Required:
Calculate the van't Hoff factor for ammonium chloride in X.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:30
Forests and meadows are often cut down to make way for farms or large number of new homes. what are some of the elements of ecosystems that are lost when plants in these areas are removed?
Answers: 2

Chemistry, 22.06.2019 06:00
Why is permeable soil best for plants that need a lot of drainage?
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2
You know the right answer?
When 70.4 g of benzamide (C7H7NO) are dissolved in 850. g of a certain mystery liquid X, the freezin...
Questions

Mathematics, 22.07.2020 19:01


Mathematics, 22.07.2020 19:01


Mathematics, 22.07.2020 19:01


History, 22.07.2020 19:01







History, 22.07.2020 19:01



Mathematics, 22.07.2020 19:01

Mathematics, 22.07.2020 19:01