
Chemistry, 25.01.2021 21:10 saraaaaaaaa20
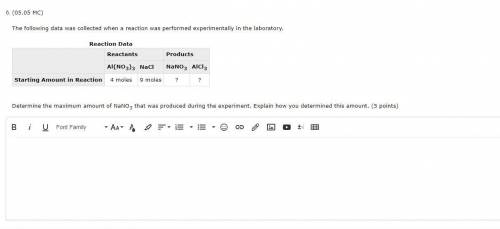
The following data was collected when a reaction was performed experimentally in the laboratory.
Reaction Data
Reactants Products
Al(NO3)3 NaCl NaNO3 AlCl3
Starting Amount in Reaction 4 moles 9 moles ? ?
Determine the maximum amount of NaNO3 that was produced during the experiment. Explain how you determined this amount. (5 points)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
If you want to create an electrical current, which situation would produce a solution capable of this
Answers: 3

Chemistry, 22.06.2019 01:30
Follow the steps provided in the simulation to add water to the graduated cylinder, select one of the three samples (copper, silver, or gold), set its mass to the values given in the statements below, and calculate its density. here is a summary of the steps required: add water by clicking and holding prepare a known volume of water button. until the desired volume of water has been added. if more than the desired volume is added, click the reset button. button and redo the procedure. a single click will add about 21.0 ml of water. to set the mass, click and hold weigh out metal button. until the desired amount of metal is added to the weighing pan. once the desired mass of the metal is added, release the button. transfer the metal to water and then click on calculate density button. to see how the density is calculated using water displacement to measure the volume of the solid. to save time you can approximate the initial volume of water to â±1 ml and the initial mass of the solid to â±1 g. for example, if you are asked to add 23 ml of water, add between 22 ml and 24 ml. which metals in each of the following sets will have equal density? check all that apply.
Answers: 1

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
You know the right answer?
The following data was collected when a reaction was performed experimentally in the laboratory.
Re...
Questions

Mathematics, 15.11.2020 02:40



Mathematics, 15.11.2020 02:40




Business, 15.11.2020 02:40



Mathematics, 15.11.2020 02:40

Computers and Technology, 15.11.2020 02:40



Social Studies, 15.11.2020 02:40



Business, 15.11.2020 02:40

Mathematics, 15.11.2020 02:40

Biology, 15.11.2020 02:40



