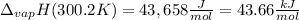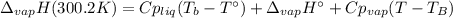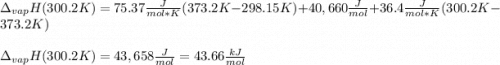Chemistry, 25.01.2021 20:40 PastelHibiscus
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The molar heat capacity of liquid water is 75.37 J K-1 mol-1 and that of gaseous water is 36.4 J K-1 mol-1. Assume that these values are independent of temperature. What is the heat of vaporization of water at 300.2 K?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3
You know the right answer?
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The molar h...
Questions


Mathematics, 16.07.2020 08:01


History, 16.07.2020 08:01




Biology, 16.07.2020 08:01

Mathematics, 16.07.2020 08:01


Business, 16.07.2020 08:01

History, 16.07.2020 08:01

Mathematics, 16.07.2020 08:01

Social Studies, 16.07.2020 08:01

Mathematics, 16.07.2020 08:01


English, 16.07.2020 08:01