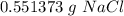1 Na2CO3(aq) + 1 CaCl2(aq) → 1 CaCO3(s) + 2 NaCl(aq) 4. Use the balanced chemical equation from the last question to solve this situation: You combine 0.5 grams of Na2CO3 with excess CaCl2. How many grams of NaCl would you expect this reaction to produce? Show all work below.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2

Chemistry, 22.06.2019 02:00
What is the maximum number of electrons that an atomic orbital can contain?
Answers: 1

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2

Chemistry, 23.06.2019 02:00
To calculate the molarity of a solution, you need to know the moles of solute and the
Answers: 2
You know the right answer?
1 Na2CO3(aq) + 1 CaCl2(aq) → 1 CaCO3(s) + 2 NaCl(aq) 4. Use the balanced chemical equation from the...
Questions



Biology, 20.09.2021 14:00





Law, 20.09.2021 14:00

Mathematics, 20.09.2021 14:00


Social Studies, 20.09.2021 14:00

English, 20.09.2021 14:00


Mathematics, 20.09.2021 14:00

Mathematics, 20.09.2021 14:00

Mathematics, 20.09.2021 14:00

Mathematics, 20.09.2021 14:00

Business, 20.09.2021 14:00


Physics, 20.09.2021 14:00

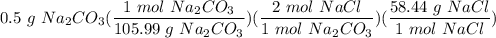 Multiply/Divide:
Multiply/Divide: