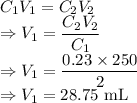Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:40
Which is a difference between molecular compounds and ionic compounds? select the correct answer below: question 5 options: molecular compounds typically form between a metal and a nonmetal, while ionic compounds typically form between nonmetals. molecular compounds result from the transfer of electrons between atoms to form ions, while ionic compounds result from the sharing of electrons between neutral atoms. molecular compounds are formed of discrete, neutral molecules, while ionic compounds are formed of large repeating arrays of opposite charges. molecular compounds have high melting points and high boiling points, while ionic
Answers: 3

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
A stock solution of Al(CH3COO)3 is available to prepare solutions that are more dilute. Calculate th...
Questions



English, 26.09.2019 14:20


Mathematics, 26.09.2019 14:20

Mathematics, 26.09.2019 14:20


Mathematics, 26.09.2019 14:20




Mathematics, 26.09.2019 14:20

Social Studies, 26.09.2019 14:20


Health, 26.09.2019 14:20

Biology, 26.09.2019 14:20



Biology, 26.09.2019 14:20

Social Studies, 26.09.2019 14:20

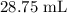
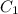 = Initial concentration = 2 M
= Initial concentration = 2 M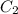 = Final concentration = 0.23 M
= Final concentration = 0.23 M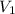 = Initial volume
= Initial volume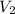 = Final volume = 250 mL
= Final volume = 250 mL