
Chemistry, 21.01.2021 14:30 hntnhtthnyt
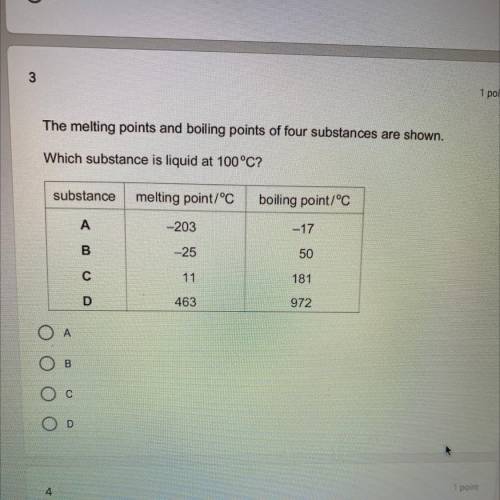
The melting points and boiling points of 4 substances are shown.
which substance is a liquid at 100 C?
A- melting( -203, boil (-17
B- melt (-25, boyl 50
C- melt 11 , boil 181
D- melt 463 boil 972

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calcium hydroxide. if 26.5 ml of base are required to neutralize 20.3 ml of the acid, what is the molarity of the hydroiodic acid solution? m hydroiodic acid
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 01:00
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3
You know the right answer?
The melting points and boiling points of 4 substances are shown.
which substance is a liquid at 100...
Questions

Business, 06.12.2020 14:00

Chemistry, 06.12.2020 14:00


Business, 06.12.2020 14:00

Mathematics, 06.12.2020 14:00

Biology, 06.12.2020 14:00

Mathematics, 06.12.2020 14:00

Mathematics, 06.12.2020 14:00



Business, 06.12.2020 14:00


History, 06.12.2020 14:00

Chemistry, 06.12.2020 14:00




Engineering, 06.12.2020 14:00

Mathematics, 06.12.2020 14:00

Mathematics, 06.12.2020 14:00



