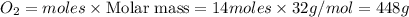Chemistry, 20.01.2021 17:20 Priskittles
Automobile catalytic converters use a platinum catalyst to reduce air pollution by changing emissions such as carbon monoxide, CO(g), into carbon dioxide, CO2(g). The uncatalyzed reaction is represented by the balanced equation below.2CO(g) O2(g) 2CO2(g) +heat. determine the mass of O2(g) required to completely react with 784g moles of CO(g) during this reaction.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
How much energy moves onto the next level, in an energy pyramid
Answers: 1

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2
You know the right answer?
Automobile catalytic converters use a platinum catalyst to reduce air pollution by changing emission...
Questions


Mathematics, 22.10.2020 18:01

Mathematics, 22.10.2020 18:01

Spanish, 22.10.2020 18:01


English, 22.10.2020 18:01




Mathematics, 22.10.2020 18:01

Mathematics, 22.10.2020 18:01

Mathematics, 22.10.2020 18:01



Mathematics, 22.10.2020 18:01

Mathematics, 22.10.2020 18:01




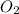 will be required to completely react with 784g moles of CO(g) during this reaction.
will be required to completely react with 784g moles of CO(g) during this reaction.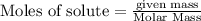
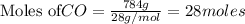
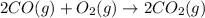
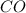 require = 1 mole of
require = 1 mole of 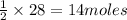 of
of