
Chemistry, 20.01.2021 01:00 sugandiwoodbine
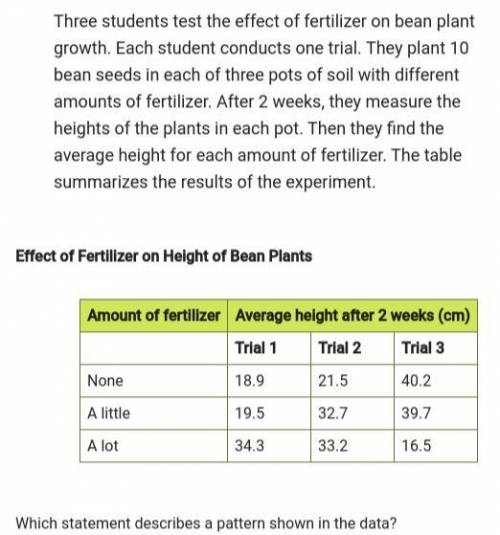
Second pic is the possible answers- science- pls help- ill give brainliest-


Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1


Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Second pic is the possible answers- science- pls help- ill give brainliest-
...
...
Questions

Mathematics, 19.01.2021 19:00

Physics, 19.01.2021 19:00


English, 19.01.2021 19:00

Computers and Technology, 19.01.2021 19:00

Mathematics, 19.01.2021 19:00


Mathematics, 19.01.2021 19:00


Arts, 19.01.2021 19:00

Social Studies, 19.01.2021 19:00


Mathematics, 19.01.2021 19:00









