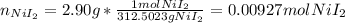Suppose 2.90g of nickel(II) iodide is dissolved in 150ml of a 0.70M aqueous solution of potassium carbonate. Calculate the final molarity of iodide anion in the solution. You can assume the volume of the solution doesn't change when the nickel(II) iodide is dissolved in it. Round your answer to 3 significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Dwayne filled a small balloon with air at 298.5 k. he put the balloon into a bucket of water, and the water level in the bucket increased by 0.54 liter. if dwayne puts the balloon into a bucket of ice water at 273.15 k and waits for the air inside the balloon come to the same temperature, what will the volume of the balloon be? assume the pressure inside the balloon doesn’t change. type the correct answer in the box. express your answer to the correct number of significant figures. the volume of the balloon at 273.15 k is liters.
Answers: 2

Chemistry, 21.06.2019 18:00
Acylinder is filled with 2.00 moles of nitrogen, 3.00 moles of argon and 5.00 moles of helium. if the gas mixture is at stp, what is the partial pressure of the argon
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1
You know the right answer?
Suppose 2.90g of nickel(II) iodide is dissolved in 150ml of a 0.70M aqueous solution of potassium ca...
Questions





Mathematics, 25.01.2022 02:10

English, 25.01.2022 02:10


Biology, 25.01.2022 02:10

Mathematics, 25.01.2022 02:10



Mathematics, 25.01.2022 02:10




Mathematics, 25.01.2022 02:10


Mathematics, 25.01.2022 02:10