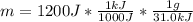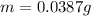Chemistry, 18.01.2021 22:40 Happypuppy1918
A certain chemical reaction releases 31.0 kj/g of heat for each gram of reactant consumed. How can you calculate what mass of reactant will produce of heat

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 02:00
What is the source of continuous heat and energy that we receive from the sun
Answers: 2
You know the right answer?
A certain chemical reaction releases 31.0 kj/g of heat for each gram of reactant consumed. How can y...
Questions



History, 02.09.2021 03:40



History, 02.09.2021 03:40



Mathematics, 02.09.2021 03:40


Mathematics, 02.09.2021 03:40



Mathematics, 02.09.2021 03:40


Mathematics, 02.09.2021 03:40


Mathematics, 02.09.2021 03:40

Mathematics, 02.09.2021 03:40