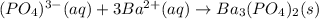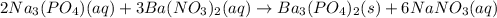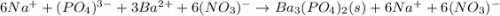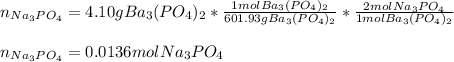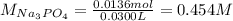Chemistry, 18.01.2021 22:00 cyaransteenberg
30.0 mL of Na3PO4(aq) is reacted with excess 2.00 M Ba(NO3)2(aq). A precipitate of Ba3(PO4)2(s) is formed. The precipitate is filtered and dried to constant mass. The mass of precipitate produced is 4.10 g. (a) Write the balanced net ionic equation of the reaction. (b) How many moles of Na3PO4 reacted

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2


Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1

Chemistry, 23.06.2019 09:10
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1
You know the right answer?
30.0 mL of Na3PO4(aq) is reacted with excess 2.00 M Ba(NO3)2(aq). A precipitate of Ba3(PO4)2(s) is f...
Questions

Mathematics, 03.07.2020 02:01


Biology, 03.07.2020 02:01

Mathematics, 03.07.2020 02:01


Mathematics, 03.07.2020 02:01



Mathematics, 03.07.2020 02:01




Mathematics, 03.07.2020 02:01

Physics, 03.07.2020 02:01


Mathematics, 03.07.2020 02:01


Computers and Technology, 03.07.2020 02:01