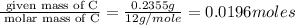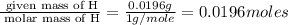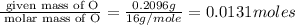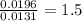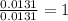Chemistry, 18.01.2021 14:00 fireemblam101ovu1gt
A 0.4647-g sample of a compound known to contain only carbon, hydrogen, and oxygen was burned in oxygen to yield 0.8635 g of CO2 and 0.1767 g of H2O. What is the empirical formula of the compound?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3

Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2

Chemistry, 23.06.2019 02:00
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1
You know the right answer?
A 0.4647-g sample of a compound known to contain only carbon, hydrogen, and oxygen was burned in oxy...
Questions

Physics, 02.02.2021 19:50


English, 02.02.2021 19:50

Chemistry, 02.02.2021 19:50

Mathematics, 02.02.2021 19:50


Mathematics, 02.02.2021 19:50



Mathematics, 02.02.2021 19:50




Mathematics, 02.02.2021 19:50



Mathematics, 02.02.2021 19:50

Mathematics, 02.02.2021 19:50

History, 02.02.2021 19:50

History, 02.02.2021 19:50

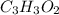 .
.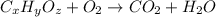
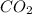 = 0.8635
= 0.8635
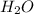 = 0.1767 g
= 0.1767 g
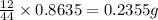 of carbon will be contained.
of carbon will be contained.
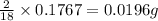 of hydrogen will be contained.
of hydrogen will be contained.