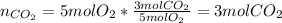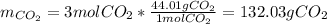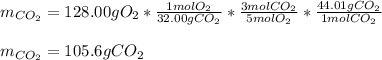A. Use the balanced equation C3H8 + 5O2 -> 3CO2 + 4H2O to answer the following questions
i. How many moles of CO2 are produced from 5 moles 02? (1 point)
ii. How many grams of CO2 are produced from 5 moles O2? (2 points)
iii how many grams of CO2 are produced from 128.00g O2? (2 points)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
A. Use the balanced equation C3H8 + 5O2 -> 3CO2 + 4H2O to answer the following questions
i. How...
Questions

Mathematics, 27.04.2021 19:10


Mathematics, 27.04.2021 19:10


Mathematics, 27.04.2021 19:10

Mathematics, 27.04.2021 19:10







Mathematics, 27.04.2021 19:10


Mathematics, 27.04.2021 19:10



Mathematics, 27.04.2021 19:10


Mathematics, 27.04.2021 19:10